 TC/PC
TC/PC
sïParadigm’s FISH TC/PC enables hospital-based pathologists to perform the Professional Component (PC) through our virtual microscopy, AI-enabled interpretation platform for hematology and solid tumors FISH testing. Generate fully customized reports branded with your facility's logo.
SCHEDULE A DEMO →
Technical Component
sïParadigm delivers complete FISH Technical processing with fully validated, AI-powered tools for superior accuracy and efficiency.
Professional Component
Facility pathologists will have full diagnostic control, with the flexibility to choose between manual and computer-assisted modes, ensuring compliance, enhanced accuracy, and user-friendliness.
Flexible Technical Excellence
You Can Always Choose Your Mode, our sïFISH platform offers dual-mode flexibility to match your workflow
Manual Digital Analysis Mode
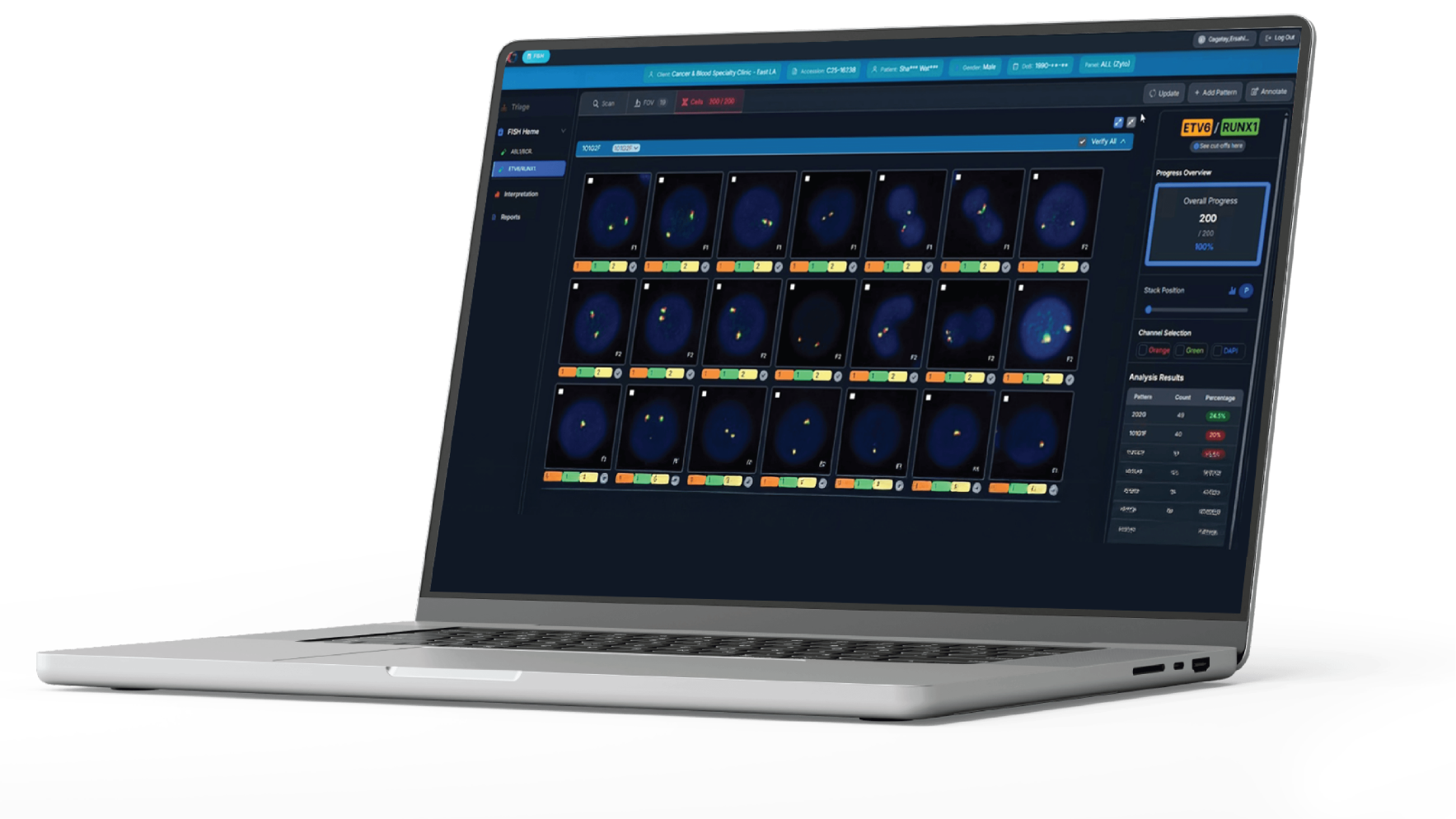
The industry’s first digital interface designed for total manual control and higher reimbursement. Analyze signal patterns using our proprietary, user-friendly AI tools to complete a full assessment in just 3 minutes per probe. You can always reflex to a full consult for complex cases.
Computer-Assisted Analysis Mode
Our best-in-class AI algorithms handle the heavy lifting—pre-screening, enumeration, and organization of signal data. This maximizes efficiency, leaving only the final professional sign-off to you.

 TC/PC
TC/PC
 TC/PC
TC/PC
Operational Excellence With Full Diagnostic Control
We provide the fastest, most efficient platform for expert interpretation, ensuring clinical excellence remains in-house.
Accelerate Your Workflow
Access our AI-Enabled Digital Interpretation Platform
Maintain Full Diagnostic Control with Flexible Client-Centric Tools
Strategic Integration and Zero Capital Investment
Access a comprehensive menu of hematologic and solid tumor FISH panels and probes, without equipment costs, additional staffing, or laboratory infrastructure investment.
How It Works
Seamless Onboarding
- Account setup.
- Expert-led siFISH training.
- Easy to follow tutorials provided through sïParadigm’s Knowledge Hub.
Technical Component
- FISH wet lab performed
- High-resolution, multi-spectrum, 3D images are processed by our advanced proprietary AI.
- Cases are pre-screened by an expert for quality assurance.
Professional Component
- Interpret findings, generate reports, and digitally sign off using integrated DocuSign.
- Download reports.
- Access a comprehensive dashboard via our Client Portal website.
We remain always your
Responsive Diagnostic Ecosystem
Offering Full Diagnostic Flexibility on Demand
Smooth, professional transition from manual to computer-assisted testing.
Convert the case to Global (full consultation) at any step.
Real-time notifications for required actions.
Customizable report templates with CPT* codes provided.
* The CPT codes provided are for information purposes only and are based on AMA guidelines. The billing party is solely responsible for correct CPT coding.
START Today …
Take Command of Your Diagnostic Workflow.
Experience the industry's most advanced AI-enabled workspace. Schedule a personalized walkthrough today.
REQUEST A DEMO →
Contact Us Directly
Our Client Services Team:
United States: 201-599-9044 or 888-599-5227, 201-599-9066 | cs@siparadigm.com
Puerto Rico: 888-782-5430, 866-369-4114 | cs.pr@siparadigm.com
Discover more.
A Model Experience
With core values rooted in service and integrity, our leadership team sets the bar high.
We consistently strive to set the model for exactly how a reference laboratory should engage with both physicians and patients.
Accredited and Certified
siParadigm is accredited by CLIA (Clinical Laboratory Improvement Amendments) and certified by CAP (College of American Pathologists).
We also hold select state licensure where required.




